Acute and Chronic Pancreatitis
Biochemistry
and Physiology of Pancreatic Exocrine Secretion
General Considerations
The
pancreas secretes 1500 to 3000 mL of isosmotic alkaline (pH >8.0) fluid per day
containing about 20 enzymes and zymogens. The pancreatic secretions provide the
enzymes needed to effect the major digestive activity of the gastrointestinal
tract and provide an optimal pH for the function of these enzymes.
Regulation of Pancreatic Secretion
The
exocrine pancreas is influenced by intimately interacting hormonal and neural
systems. Gastric acid is the stimulus for the release of secretin, a
peptide with 27 amino acids. Sensitive radioimmunoassay studies for secretin
suggest that the pH threshold for its release from the duodenum and jejunum is
4.5. Secretin stimulates the secretion of pancreatic juice rich in water and
electrolytes. Release of cholecystokinin (CCK) from the duodenum and
jejunum is largely triggered by long-chain fatty acids, certain essential amino
acids (tryptophan, phenylalanine, valine, methionine), and gastric acid itself.
CCK evokes an enzyme-rich secretion from the pancreas. Gastrin, although
it has the same terminal tetrapeptide as CCK, is a weak stimulus for pancreatic
enzyme output. The parasympathetic nervous system (via the vagus nerve)
exerts significant control over pancreatic secretion. Secretion evoked by
secretin and CCK depends on permissive roles of vagal afferent and efferent
pathways. This is particularly true for enzyme secretion, whereas water and
bicarbonate secretion is heavily dependent on the hormonal effects of secretin
and CCK. Also, vagal stimulation effects the release of vasoactive intestinal
peptide (VIP), a secretin agonist. Bile salts also stimulate pancreatic
secretion, thereby integrating the functions of the biliary tract, pancreas,
and small intestine.
Somatostatin
acts on multiple sites to induce inhibition of pancreatic secretion. The
appropriate roles of other peptides, such as peptide YY, pancreastatin,
gastrin-releasing peptide, pituitary adenylate cyclase-activating polypeptide,
calcitonin gene-related peptide, and galanin are still being defined. Nitric
oxide is an important neurotransmitter in the regulation of pancreatic exocrine
secretion, although its mechanism of action has not been fully elucidated.
Water and Electrolyte Secretion
Although
sodium, potassium, chloride, calcium, zinc, phosphate, and sulfate are found in
pancreatic secretions, bicarbonate is the ion of primary physiologic
importance. In the acini and in the ducts, secretin causes the cells to add
water and bicarbonate to the fluid. In the ducts, an exchange occurs between
bicarbonate and chloride. There is a good correlation between the maximal
bicarbonate output after stimulation with secretin and the pancreatic mass. The
bicarbonate output of 120 to 300 mmol/d helps neutralize gastric acid and
creates the appropriate pH for the activity of the pancreatic enzymes.
Enzyme Secretion
The
pancreas secretes amylolytic, lipolytic, and proteolytic enzymes. Amylolytic
enzymes, such as amylase, hydrolyze starch to oligosaccharides and to the
disaccharide maltose. The lipolytic enzymes include lipase,
phospholipase A, and cholesterol esterase. Bile salts inhibit lipase in
isolation; but colipase, another constituent of pancreatic secretion, binds to
lipase and prevents this inhibition. Bile salts activate phospholipase A
and cholesterol esterase. Proteolytic enzymes include endopeptidases
(trypsin, chymotrypsin), which act on internal peptide bonds of proteins and
polypeptides; exopeptidases (carboxypeptidases, aminopeptidases), which
act on the free carboxyl- and amino-terminal ends of peptides, respectively;
and elastase. The proteolytic enzymes are secreted as inactive precursors
(zymogens). Ribonucleases (deoxyribonucleases, ribonuclease) are also secreted.
Although pancreatic enzymes usually are secreted in parallel, nonparallel
secretion can occur as a result of exocytosis from heterogeneous sources in the
pancreas. Enterokinase, an enzyme found in the duodenal mucosa, cleaves
the lysine-isoleucine bond of trypsinogen to form trypsin. Trypsin then
activates the other proteolytic zymogens in a cascade phenomenon. All
pancreatic enzymes have pH optima in the alkaline range.
Autoprotection of the Pancreas
Autodigestion
of the pancreas is prevented by the packaging of proteases in precursor form
and by the synthesis of protease inhibitors. These protease inhibitors are
found in the acinar cell, the pancreatic secretions, and the alpha1-
and alpha2-globulin fractions of plasma.
Exocrine-Endocrine Relationships
Insulin
appears to be needed locally for secretin and CCK to promote exocrine
secretion; thus, it acts in a permissive role for these two hormones.
Enteropancreatic Axis and Feedback Inhibition
Pancreatic
enzyme secretion is controlled, at least in part, by a negative feedback
mechanism induced by the presence of active serine proteases in the duodenum.
To illustrate, perfusion of the duodenal lumen with phenylalanine causes a
prompt increase in plasma CCK levels as well as increased secretion of
chymotrypsin. However, simultaneous perfusion with trypsin blunts both
responses. Conversely, perfusion of the duodenal lumen with protease inhibitors
actually leads to enzyme hypersecretion. The available evidence supports the
concept that the duodenum contains a peptide called CCK releasing factor
(CCK-RF) that is involved in stimulating CCK release. Two peptides, luminal
CCK-RF and diazepam-binding inhibitor, have been found that may be the CCK-RF.
Serine proteases inhibit pancreatic secretion by acting on CCK-RF. It appears
that serine proteases inhibit pancreatic secretion by acting on a CCK-releasing
peptide in the lumen of the small intestine.
Acute Pancreatitis
General Considerations
Pancreatic
inflammatory disease may be classified as (1) acute pancreatitis and (2)
chronic pancreatitis. The pathologic spectrum of acute pancreatitis varies from
edematous pancreatitis, which is usually a mild and self-limited
disorder, to necrotizing pancreatitis, in which the degree of pancreatic
necrosis correlates with the severity of the attack and its systemic
manifestations. The term hemorrhagic pancreatitis is less meaningful in
a clinical sense because variable amounts of interstitial hemorrhage can be
found in pancreatitis as well as in other disorders such as pancreatic trauma,
pancreatic carcinoma, and severe congestive heart failure.
The
incidence of pancreatitis varies in different countries and depends on cause,
e.g., alcohol, gallstones, metabolic factors, and drugs (Table 304-1). In the
Table 304-1: Causes of Acute Pancreatitis
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Etiology and Pathogenesis
There
are many causes of acute pancreatitis (Table 304-1), but the mechanisms by
which these conditions trigger pancreatic inflammation have not been
identified. Alcoholic patients with pancreatitis may represent a special
subset, since most alcoholics do not develop pancreatitis. The list of
identifiable causes is growing, and it is likely that pancreatitis related to
viral infections, drugs, and as yet undefined factors is more common than
heretofore recognized.
Approximately
2 to 5% of cases of acute pancreatitis are drug-related (Table 304-1). Drugs
cause pancreatitis either by a hypersensitivity reaction or by the generation
of a toxic metabolite, although in some cases it is not clear which of these
mechanisms is operative.
Autodigestion
is one pathogenetic theory, according to which pancreatitis results when
proteolytic enzymes (e.g., trypsinogen, chymotrypsinogen, proelastase, and
phospholipase A) are activated in the pancreas rather than in the intestinal
lumen. A number of factors (e.g., endotoxins, exotoxins, viral infections,
ischemia, anoxia, and direct trauma) are believed to activate these proenzymes.
Activated proteolytic enzymes, especially trypsin, not only digest pancreatic
and peripancreatic tissues but also can activate other enzymes, such as elastase
and phospholipase. The active enzymes then digest cellular membranes and cause
proteolysis, edema, interstitial hemorrhage, vascular damage, coagulation
necrosis, fat necrosis, and parenchymal cell necrosis. Cellular injury and
death result in the liberation of activated enzymes. In addition, activation
and release of bradykinin peptides and vasoactive substances (e.g., histamine)
are believed to produce vasodilation, increased vascular permeability, and
edema. Thus, a cascade of events culminates in the development of acute
necrotizing pancreatitis.
The
autodigestion theory has largely eclipsed two older theories. First, according
to the "common channel" theory, the existence of a common anatomic
channel for pancreatic secretions and bile permits reflux of bile into the
pancreatic duct, which results in activation of pancreatic enzymes. (Actually,
a common channel with free communication between the common bile duct and the
main pancreatic duct is infrequently encountered.) The second theory is that obstruction
and hypersecretion are pivotal in the development of pancreatitis. Obstruction
of the main pancreatic duct, however, produces pancreatic edema but generally
not pancreatitis.
A
recent hypothesis to explain the intrapancreatic activation of zymogens is that
they become activated by lysosomal hydrolases in the pancreatic acinar
cell itself. In two different types of experimental pancreatitis, it has been
demonstrated that digestive enzymes and lysosomal hydrolases become admixed; as
a result, the latter can activate the former in the acinar cell. In vitro,
lysosomal enzymes such as cathepsin B can activate trypsinogen, and trypsin can
activate the other protease precursors. It is still not clear, however, whether
the human acinar cell can provide the pH (about 3.0) necessary for activation
of trypsinogen by lysosomal hydrolases. It is now believed that
ischemia/hypoperfusion can alone result in activation of trypsinogen and
pancreatic injury.
Clinical Features
Abdominal pain is the major symptom of acute pancreatitis.
Pain may vary from a mild and tolerable discomfort to severe, constant, and
incapacitating distress. Characteristically, the pain, which is steady and
boring in character, is located in the epigastrium and periumbilical region and
often radiates to the back as well as to the chest, flanks, and lower abdomen.
The pain is frequently more intense when the patient is supine, and patients
often obtain relief by sitting with the trunk flexed and knees drawn up.
Nausea, vomiting, and abdominal distention due to gastric and intestinal
hypomotility and chemical peritonitis are also frequent complaints.
Physical examination frequently reveals a distressed and anxious
patient. Low-grade fever, tachycardia, and hypotension are fairly common. Shock
is not unusual and may result from (1) hypovolemia secondary to exudation of
blood and plasma proteins into the retroperitoneal space (a
"retroperitoneal burn"); (2) increased formation and release of kinin
peptides, which cause vasodilation and increased vascular permeability; and (3)
systemic effects of proteolytic and lipolytic enzymes released into the
circulation. Jaundice occurs infrequently; when present, it usually is due to
edema of the head of the pancreas with compression of the intrapancreatic portion
of the common bile duct. Erythematous skin nodules due to subcutaneous fat
necrosis may occur. In 10 to 20% of patients, there are pulmonary findings,
including basilar rales, atelectasis, and pleural effusion, the latter most
frequently left-sided. Abdominal tenderness and muscle rigidity are present to
a variable degree, but, compared with the intense pain, these signs may be
unimpressive. Bowel sounds are usually diminished or absent. A pancreatic
pseudocyst may be palpable in the upper abdomen. A faint blue discoloration
around the umbilicus (Cullen's sign) may occur as the result of hemoperitoneum,
and a blue-red-purple or green-brown discoloration of the flanks (Turner's
sign) reflects tissue catabolism of hemoglobin. The latter two findings, which
are uncommon, indicate the presence of a severe necrotizing pancreatitis.
Laboratory Data
The
diagnosis of acute pancreatitis is usually established by the detection of an
increased level of serum amylase. Values threefold or more above normal
virtually clinch the diagnosis if overt salivary gland disease and gut
perforation or infarction are excluded. However, there appears to be no
definite correlation between the severity of pancreatitis and the degree of
serum amylase elevation. After 48 to 72 h, even with continuing evidence of
pancreatitis, total serum amylase values tend to return to normal. However,
pancreatic isoamylase and lipase levels may remain elevated for 7 to 14 days.
It will be recalled that amylase elevations in serum and urine occur in many
conditions other than pancreatitis (see Table 303-2). Importantly, patients
with acidemia (arterial pH ![]() 7.32)
may have spurious elevations in serum amylase. In one study, 12 of 33 patients
with acidemia had elevated serum amylase, but only 1 had an elevated lipase
value; in 9, salivary-type amylase was the predominant serum isoamylase. This
finding explains why patients with diabetic ketoacidosis may have marked
elevations in serum amylase without any other evidence of acute pancreatitis.
Serum lipase activity increases in parallel with amylase activity, and
measurement of both enzymes increases the diagnostic yield. An elevated serum
lipase or trypsin value is usually diagnostic of acute pancreatitis; these
tests are especially helpful in patients with nonpancreatic causes of
hyperamylasemia (see Table 303-2). Markedly increased levels of peritoneal or
pleural fluid amylase [>1500 nmol/L (> 5000 U/dL)] are also helpful, if
present, in establishing the diagnosis.
7.32)
may have spurious elevations in serum amylase. In one study, 12 of 33 patients
with acidemia had elevated serum amylase, but only 1 had an elevated lipase
value; in 9, salivary-type amylase was the predominant serum isoamylase. This
finding explains why patients with diabetic ketoacidosis may have marked
elevations in serum amylase without any other evidence of acute pancreatitis.
Serum lipase activity increases in parallel with amylase activity, and
measurement of both enzymes increases the diagnostic yield. An elevated serum
lipase or trypsin value is usually diagnostic of acute pancreatitis; these
tests are especially helpful in patients with nonpancreatic causes of
hyperamylasemia (see Table 303-2). Markedly increased levels of peritoneal or
pleural fluid amylase [>1500 nmol/L (> 5000 U/dL)] are also helpful, if
present, in establishing the diagnosis.
Leukocytosis (15,000 to 20,000 leukocytes per microliter)
occurs frequently. Patients with more severe disease may show hemoconcentration
with hematocrit values exceeding 50% because of loss of plasma into the
retroperitoneal space and peritoneal cavity. Hyperglycemia is common and
is due to multiple factors, including decreased insulin release, increased
glucagon release, and an increased output of adrenal glucocorticoids and
catecholamines. Hypocalcemia occurs in approximately 25% of patients,
and its pathogenesis is incompletely understood. Although earlier studies
suggested that the response of the parathyroid gland to a decrease in serum
calcium is impaired, subsequent observations have failed to confirm this idea.
Intraperitoneal saponification of calcium by fatty acids in areas of fat
necrosis occurs occasionally, with large amounts (up to 6.0 g) dissolved or
suspended in ascitic fluid. Such "soap formation" also may be
significant in patients with pancreatitis, mild hypocalcemia, and little or no
obvious ascites. Hyperbilirubinemia [serum bilirubin >68 ![]() mol/L
(> 4.0 mg/dL)] occurs in approximately 10% of patients. However, jaundice is
transient, and serum bilirubin levels return to normal in 4 to 7 days. Serum
alkaline phosphatase and aspartate aminotransferase (AST) levels are also
transiently elevated and parallel serum bilirubin values. Markedly elevated
serum lactic dehydrogenase (LDH) levels [>8.5
mol/L
(> 4.0 mg/dL)] occurs in approximately 10% of patients. However, jaundice is
transient, and serum bilirubin levels return to normal in 4 to 7 days. Serum
alkaline phosphatase and aspartate aminotransferase (AST) levels are also
transiently elevated and parallel serum bilirubin values. Markedly elevated
serum lactic dehydrogenase (LDH) levels [>8.5 ![]() mol/L
(> 500 U/dL)] suggest a poor prognosis. Serum albumin is decreased to
mol/L
(> 500 U/dL)] suggest a poor prognosis. Serum albumin is decreased to ![]() 30
g/L (
30
g/L (![]() 3.0
g/dL) in about 10% of patients; this sign is associated with more severe
pancreatitis and a higher mortality rate (Table 304-2). Hypertriglyceridemia
occurs in 15 to 20% of patients, and serum amylase levels in these individuals
are often spuriously normal (Chap. 303). Most patients with
hypertriglyceridemia and pancreatitis, when subsequently examined, show evidence
of an underlying derangement in lipid metabolism which probably antedated the
pancreatitis (see below). Approximately 25% of patients have hypoxemia
(arterial PO2
3.0
g/dL) in about 10% of patients; this sign is associated with more severe
pancreatitis and a higher mortality rate (Table 304-2). Hypertriglyceridemia
occurs in 15 to 20% of patients, and serum amylase levels in these individuals
are often spuriously normal (Chap. 303). Most patients with
hypertriglyceridemia and pancreatitis, when subsequently examined, show evidence
of an underlying derangement in lipid metabolism which probably antedated the
pancreatitis (see below). Approximately 25% of patients have hypoxemia
(arterial PO2 ![]() 60
mmHg), which may herald the onset of acute respiratory distress syndrome.
Finally, the electrocardiogram is occasionally abnormal in acute pancreatitis
with ST-segment and T-wave abnormalities simulating myocardial ischemia.
60
mmHg), which may herald the onset of acute respiratory distress syndrome.
Finally, the electrocardiogram is occasionally abnormal in acute pancreatitis
with ST-segment and T-wave abnormalities simulating myocardial ischemia.
Table 304-2: Factors That Adversely Affect
Survival in Acute Pancreatitis
|
Although
one or more radiologic abnormalities are found in over 50% of patients, the findings
are inconstant and nonspecific. The chief value of conventional x-rays [chest
films; kidney, ureter, and bladder (KUB) studies] in acute pancreatitis is to
help exclude other diagnoses, especially a perforated viscus. Upper
gastrointestinal tract x-rays have been superseded by ultrasonography and
computed tomography (CT). A CT scan may confirm the clinical impression of
acute pancreatitis even in the face of normal serum amylase levels.
Importantly, CT is quite helpful in indicating the severity of acute
pancreatitis and the risk of morbidity and mortality (see below). Sonography
and radionuclide scanning [N-p-isopropylacetanilide-iminodiacetic
acid (PIPIDA) scan; hepatic 2,6-dimethyliminodiacetic acid (HIDA) scan] are useful
in acute pancreatitis to evaluate the gallbladder and biliary tree. Radiologic
303 and 303-1.
Diagnosis
Any
severe acute pain in the abdomen or back should suggest acute pancreatitis. The
diagnosis is usually entertained when a patient with a possible predisposition
to pancreatitis presents with severe and constant abdominal pain, nausea,
emesis, fever, tachycardia, and abnormal findings on abdominal examination.
Laboratory studies frequently reveal leukocytosis, an abnormal appearance on
x-rays of the abdomen and chest, hypocalcemia, and hyperglycemia. The diagnosis
is usually confirmed by the finding of an elevated level of serum amylase
and/or lipase. Not all the above features have to be present for the diagnosis
to be established.
The
differential diagnosis should include the following disorders: (1)
perforated viscus, especially peptic ulcer; (2) acute cholecystitis and biliary
colic; (3) acute intestinal obstruction; (4) mesenteric vascular occlusion; (5)
renal colic; (6) myocardial infarction; (7) dissecting aortic aneurysm; (8)
connective tissue disorders with vasculitis; (9) pneumonia; and (10) diabetic
ketoacidosis. A penetrating duodenal ulcer usually can be identified by upper
gastrointestinal x-rays and/or endoscopy. A perforated duodenal ulcer is
readily diagnosed by the presence of free intraperitoneal air. It may be
difficult to differentiate acute cholecystitis from acute pancreatitis, since
an elevated serum amylase may be found in both disorders. Pain of biliary tract
origin is more right-sided and gradual in onset, and ileus is usually absent;
sonography and radionuclide scanning are helpful in establishing the diagnosis
of cholelithiasis and cholecystitis. Intestinal obstruction due to mechanical
factors can be differentiated from pancreatitis by the history of colicky pain,
findings on abdominal examination, and x-rays of the abdomen showing changes
characteristic of mechanical obstruction. Acute mesenteric vascular occlusion
is usually evident in elderly debilitated patients with brisk leukocytosis,
abdominal distention, and bloody diarrhea, in whom paracentesis shows
sanguineous fluid and arteriography shows vascular occlusion. Serum as well as
peritoneal fluid amylase levels are increased, however, in patients with
intestinal infarction. Systemic lupus erythematosus and polyarteritis nodosa
may be confused with pancreatitis, especially since pancreatitis may develop as
a complication of these diseases. Diabetic ketoacidosis is often accompanied by
abdominal pain and elevated total serum amylase levels, thus closely mimicking
acute pancreatitis. However, the serum lipase and pancreatic isoamylase levels
are not elevated in diabetic ketoacidosis.
Course of the Disease and Complications
It
is important to identify patients with acute pancreatitis who have an increased
risk of dying. Ranson and Imrie have used multiple prognostic criteria and have
demonstrated that there is an increased mortality rate when three or more risk
factors are identifiable either at the time of admission to the hospital or
during the initial 48 h of hospitalization (Table 304-2). Recent studies
indicate that obesity is a major risk factor for severe pancreatitis,
presumably because the increased deposits of peripancreatic fat in such
patients may predispose them to more extensive pancreatic and peripancreatic
necrosis. The acute physiology and chronic health evaluation scoring system
(APACHE II) uses the worst values of 12 physiologic measurements plus age and
previous health status and provides a good description of illness severity for
a wide range of common diseases; this score also correlates with outcome.
Prospective studies have compared APACHE II with multiple prognostic criteria,
i.e., Ranson and Imrie scores, in predicting the severity of acute pancreatitis.
On admission, APACHE II identified approximately two-thirds of severe attacks,
and after 48 h, the prognostic accuracy of APACHE II is comparable with that of
Ranson and Imrie's scoring system. The drawbacks of APACHE II are (1) its
complexity, (2) the requirement of a computer for scoring, and (3)
standardization regarding peak values and cutoff scores. McMahon and colleagues
have shown that the presence of a "toxic broth" or dark (hemorrhagic)
fluid in abdominal pancreatitis is also an important prognostic indicator in
acute pancreatitis. These multiple-factor scoring systems are difficult to use
and have not been embraced consistently by clinicians. There is a great need
for a reliable, simple biochemical test that consistently predicts outcome in
patients with acute pancreatitis. Three candidate markers that show great
promise are C-reactive protein, serum granulocyte elastase, and urinary
trypsinogen activation peptide (TAP). The key indicators of a severe attack of
acute pancreatitis are also listed in Table 304-2. Importantly, the presence of
any one of these factors is associated with an increased risk of complications,
and the presence of any two, with a 20 to 30% mortality rate. The high
mortality rate of such severely ill patients is due in large part to infection
and warrants intensive radiologic intervention and monitoring and/or a
combination of radiologic and surgical means, as discussed in detail below.
The
local and systemic complications of acute pancreatitis are listed in Table
304-3. In the first 2 to 3 weeks after pancreatitis patients frequently develop
an inflammatory mass, which may be due to pancreatic necrosis (with or without
infection) or may represent an abscess or pseudocyst (see below). Systemic
complications include pulmonary, cardiovascular, hematologic, renal, metabolic,
and central nervous system abnormalities. Pancreatitis and hypertriglyceridemia
constitute an association in which cause and effect remain incompletely
understood. However, several reasonable conclusions can be drawn. First,
hypertriglyceridemia can precede and apparently cause pancreatitis. Second, the
vast majority (>80%) of patients with acute pancreatitis do not have
hypertriglyceridemia. Third, almost all patients with pancreatitis and
hypertriglyceridemia have preexisting abnormalities in lipoprotein metabolism.
Fourth, many of the patients with this association have persistent
hypertriglyceridemia after recovery from pancreatitis and are prone to
recurrent episodes of pancreatitis. Fifth, any factor (e.g., drugs or alcohol)
that causes an abrupt increase in serum triglycerides to levels greater than 11
mmol/L (1000 mg/dL) can precipitate a bout of pancreatitis that can be
associated with significant complications and even become fulminant. To avert
the risk of triggering pancreatitis, a fasting serum triglyceride measurement
should be obtained before estrogen replacement therapy is begun in
postmenopausal women. Fasting levels less than 300 mg/dL pose no risk, whereas
levels greater than 750 mg/dL are associated with a high probability of
developing pancreatitis. Finally, patients with a deficiency of apolipoprotein
CII have an increased incidence of pancreatitis; apolipoprotein CII activates
lipoprotein lipase, which is important in clearing chylomicrons from the
bloodstream.
Table 304-3: Complications of Acute Pancreatitis
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Purtscher's retinopathy, a relatively unusual complication, is
manifested by a sudden and severe loss of vision in a patient with acute
pancreatitis. It is characterized by a peculiar funduscopic appearance with
cotton-wool spots and hemorrhages confined to an area limited by the optic disk
and macula; it is believed to be due to occlusion of the posterior retinal
artery with aggregated granulocytes.
The
two most common causes of acute pancreatitis are alcoholism and biliary tract
disease; other causes are listed in Table 304-1. The risk of acute pancreatitis
in patients with at least one gallstone smaller than 5 mm in diameter is
fourfold greater than that in patients with larger stones. However, after a
conventional workup, a specific cause is not identified in about 30% of patients.
It is important to note that ultrasound examinations fail to detect gallstones,
especially microlithiasis and/or sludge, in 4 to 7% of patients. In one series
of 31 patients diagnosed initially as having idiopathic acute pancreatitis, 23
were found to have occult gallstone disease. Thus, approximately two-thirds of
patients with recurrent acute pancreatitis without an obvious cause actually
have occult gallstone disease due to microlithiasis. Examination of duodenal
aspirates in such cases often reveals cholesterol crystals, which confirm the
diagnosis. Other diseases of the biliary tree and pancreatic ducts that can
cause acute pancreatitis include choledochocele, ampullary tumors, pancreas
divisum, and pancreatic duct stones, stricture, and tumor. Approximately 2% of
patients with pancreatic carcinoma present with acute pancreatitis.
Pancreatitis in Patients with AIDS
The
incidence of acute pancreatitis is increased in patients with AIDS for two
reasons: (1) the high incidence of infections involving the pancreas, such as
infections with cytomegalovirus, Cryptosporidium, and the Mycobacterium
avium complex; and (2) the frequent use by patients with AIDS of
medications such as didanosine, pentamidine, and trimethoprim-sulfamethoxazole
(Chap. 309).
![]() Treatment
Treatment
In
most patients (approximately 85 to 90%) with acute pancreatitis, the disease is
self-limited and subsides spontaneously, usually within 3 to 7 days after
treatment is instituted. Conventional measures include (1) analgesics for pain,
(2) intravenous fluids and colloids to maintain normal intravascular volume,
(3) no oral alimentation, and (4) nasogastric suction to decrease gastrin release
from the stomach and prevent gastric contents from entering the duodenum.
Recent controlled trials, however, have shown that nasogastric suction offers
no clear-cut advantages in the treatment of mild to moderately severe acute
pancreatitis. Its use, therefore, must be considered elective rather than
mandatory.
It
has been demonstrated that CCK-stimulated pancreatic secretion is almost
abolished in four different experimental models of acute pancreatitis. This
finding probably explains why drugs to block pancreatic secretion in acute
pancreatitis have failed to have any therapeutic benefit. For this and other
reasons, anticholinergic drugs are not indicated in acute pancreatitis. In
addition to nasogastric suction and anticholinergic drugs, other therapies
designed to "rest the pancreas" by inhibiting pancreatic secretion
have not changed the course of the disease. Although antibiotics have been used
in the treatment of acute pancreatitis, randomized, prospective trials have
shown no benefit from their use in acute pancreatitis of mild to moderate
severity.
However,
current experimental evidence favors the use of prophylactic antibiotics in
severe acute pancreatitis. Results of four contemporary randomized clinical
trials restricted to patients with prognostically severe acute pancreatitis
have demonstrated an improved outcome, i.e., reduced rate of infection and/or
mortality, associated with the antibiotic treatment. The carbapenem group of
antibiotics, including imipenem, has a very broad spectrum including activity
against Pseudomonas, Staphylococcus, and Enterococcus; and
these agents penetrate well into pancreatic tissue. Furthermore, because
secondary infection of necrotic pancreatic tissue (abscess, pseudocyst or obstructed
biliary passages, ascending cholangitis complicating choledocholithiasis)
contributes to many of the late deaths from pancreatitis, appropriate
antibiotic therapy of established infections is quite important.
Several
other drugs have been evaluated by prospective controlled trials and found
ineffective in the treatment of acute pancreatitis. The list, by no means
complete, includes glucagon, H2 blockers, protease inhibitors such
as aprotinin, glucocorticoids, calcitonin, nonsteroidal anti-inflammataory
drugs (NSAIDs) and lexiplafant, a platelet-activating factor inhibitor. A
recent meta-analysis of somatostatin, ocreotide, and the antiprotease gabexate
methylate in therapy of acute pancreatitis suggested (1) a reduced mortality
rate but no change in complications with octreotide, and (2) no effect on the
mortality rate but reduced pancreatic damage with gabexate.
Intraabdominal
Candida infection during acute necrotizing pancreatitis is increasing in
frequency and is associated with an increased mortality rate. In one
representatitve trial, intraabdominal Candida infection was found in 13
of 37 cases and was associated with a mortality rate fourfold greater than that
associated with intraabdominal bacterial infection alone. Given the impact of Candida
infection on the mortality rate in acute necrotizing pancreatitis and the
apparent benefit of prophylactic chemotherapy, these data suggest earlier use
of fungicides.
A
CT scan, especially a contrast-enhanced dynamic CT (CECT) scan, provides
valuable information on the severity and prognosis of acute pancreatitis (Fig.
304-1 and Table 304-4). In particular, a CECT scan allows estimation of the
presence and extent of pancreatic necrosis. Recent studies suggest that the
likelihood of prolonged pancreatitis or a serious complication is negligible
when the CT severity index is 1 or 2 and low with scores of 3 to 6. However,
patients with scores of 7 to 10 had a 92% morbidity rate and a 17% mortality
rate. Necrosis is present in 20 to 30% of patients. Those with necrosis have a
morbidity rate >20%, whereas those without necrosis have a morbidity rate
<10% and a negligible mortality rate. A CECT scan is indicated in patients
with three or more of Ranson's signs, in all seriously ill patients, and in
patients who show evidence of clinical deterioration. The patient with mild to
moderate pancreatitis usually requires treatment with intravenous fluids,
fasting, and possibly nasogastric suction for 2 to 4 days. A clear liquid diet
is frequently started on the third to sixth day, and a regular diet by the
fifth to seventh day. The patient with unremitting fulminant pancreatitis
usually requires inordinate amounts of fluid and close attention to
complications such as cardiovascular collapse, respiratory insufficiency, and
pancreatic infection. The latter should be managed by a combination of
radiologic and surgical means (see below). While earlier uncontrolled studies
suggested that peritoneal lavage through a percutaneous dialysis
catheter was helpful in severe pancreatitis, subsequent studies indicate that
this treatment does not influence the outcome of such attacks. Aggressive
surgical pancreatic debridement (necrosectomy) should be undertaken soon after
confirmation of the presence of infected necrosis, and multiple operations may
be required. Since the mortality rate from sterile acute necrotizing
pancreatitis is approximately 10%, laparotomy with adequate drainage and
removal of necrotic tissue should be considered if conventional therapy does
not halt the patient's deterioration. The use of parenteral nutrition makes it
possible to give nutritional support to patients with severe, acute, or
protracted pancreatitis who are unable to eat normally. Patients with severe
gallstone-induced pancreatitis may improve dramatically if papillotomy is
carried out within the first 36 to 72 h of the attack. Studies indicate that
only those patients with gallstone pancreatitis who are in the very severe
group should be considered for urgent endoscopic retrograde
cholangiopancreatography (ERCP). Finally, the treatment for patients with
hypertriglyceridemia-associated pancreatitis includes (1) weight loss to ideal
weight, (2) a lipid-restricted diet, (3) exercise, (4) avoidance of alcohol and
of drugs that can elevate serum triglycerides (i.e., estrogens, vitamin A,
thiazides, and beta-blockers), and (5) control of diabetes.
|
|
|
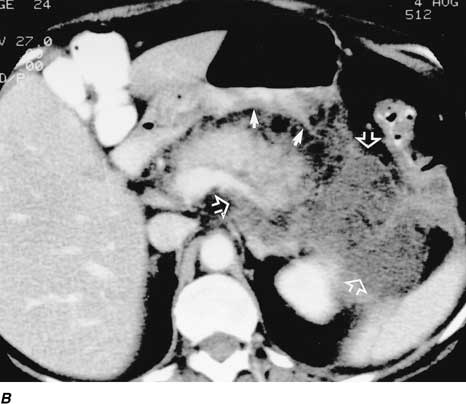
Figure 304-1: Acute pancreatitis: CT evolution. A.
Contrast-enhanced CT scan of the abdomen performed on admission of a patient
with clinical evidence of acute pancreatitis. Note the mildly decreased density
of the body of the pancreas to the left of the midline (arrow). There
are a few linear strands in the peripancreatic fat, suggesting inflammation (open
arrows). A small amount of fluid is seen in the anterior pararenal space (arrowhead).
B. Nine days after admission, there is a marked worsening with severe
inflammation of the pancreas evidenced by anterior displacement of the
posterior gastric wall (arrows), increased inflammation of the
peripancreatic fat, and increased pancreatic effusion in the anterior perirenal
space and around the splenic vein (open arrows).
Table 304-4: Severity Index in Acute
Pancreatitis
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Infected Pancreatic Necrosis, Abscess, and
Pseudocyst
Infected
pancreatic necrosis should be differentiated from pancreatic abscess. The
former is a diffuse infection of an acutely inflamed, necrotic pancreas
occurring in the first 1 to 2 weeks after the onset of pancreatitis. In
contrast, a pancreatic abscess is an ill-defined, liquid collection of pus that
evolves over a longer period, often 4 to 6 weeks. It tends to be less
life-threatening and is associated with a lower rate of surgical mortality.
Infected pancreatic necrosis should be treated by surgical debridement because
the solid component of the infected pancreas is not amenable to effective
radiologically guided percutaneous evacuation. Pancreatic abscess can be
treated surgically or, in selected cases, by percutaneous drainage. The necrotic
pancreas becomes secondarily infected in 40 to 60% of patients, most frequently
with gram-negative bacteria of alimentary origin. Whether infection occurs
depends on several factors, including the extent of pancreatic and
peripancreatic necrosis, the degree of pancreatic ischemia and hypoperfusion,
and the presence of organ or multiorgan failure.
The
early diagnosis of pancreatic infection can be accomplished by CT-guided needle
aspiration. In one study, 60 patients, representing 5% of all admissions for
acute pancreatitis, were suspected of harboring a pancreatic infection on the
basis of fever, leukocytosis, and an abnormal CT scan (pseudocyst or
extrapancreatic fluid collection). Importantly, 60% of these patients had a
pancreatic infection, and 55% of these infections developed in the first 2
weeks. These findings suggest that only guided aspiration can reliably
distinguish sterile from infected pancreatic necrosis. The following are
guidelines for patients meeting the above selection criteria: (1) Pseudocysts
should be aspirated promptly, because more than half may be infected; (2)
extrapancreatic fluid collections need not be aspirated promptly, because most
are sterile; (3) if a necrotic pancreas is found initially to be sterile but
fever and leukocytosis persist, several days of observation should be allowed
to pass before reaspiration is considered, as clinical improvement frequently
occurs; and (4) if fever and leukocytosis recur after an interval of
well-being, reaspiration should be considered.
Severe
pancreatitis with the presence of three or more risk factors, postoperative
pancreatitis, early oral feeding, early laparotomy, and perhaps injudicious use
of antibiotics predispose to the development of pancreatic abscess, which
occurs in 3 to 4% of patients with acute pancreatitis. Pancreatic abscess also
may develop because of a communication between a pseudocyst and the colon,
inadequate surgical drainage of a pseudocyst, or needling of a pseudocyst. The
characteristic signs of abscess are fever, leukocytosis, ileus, and rapid
deterioration in a patient previously recovering from pancreatitis. Sometimes,
however, the only manifestations are persistent fever and signs of continuing
pancreatic inflammation. Drainage of pancreatic abscesses by percutaneous
catheter techniques, using CT guidance, has been only moderately successful
(resolution in 50 to 60% of patients). Accordingly, laparotomy with radical
sump drainage and possibly resection of necrotic tissue is usually required,
because the mortality rate for undrained pancreatic abscess approaches 100%.
Multiple abscesses are common, and reoperation is frequently necessary.
Pseudocysts of the pancreas are collections of tissue,
fluid, debris, pancreatic enzymes, and blood which develop over a period of 1
to 4 weeks after the onset of acute pancreatitis; they form in approximately
15% of patients with acute pancreatitis. In contrast to true cysts, pseudocysts
do not have an epithelial lining; their walls consist of necrotic tissue,
granulation tissue, and fibrous tissue. Disruption of the pancreatic ductal
system is common. However, the subsequent course of this disruption varies
widely, ranging from spontaneous healing to continuous leakage of pancreatic
juice, which results in tense ascites. Pseudocysts are preceded by pancreatitis
in 90% of cases and by trauma in 10%. Approximately 85% are located in the body
or tail of the pancreas and 15% in the head. Some patients have two or more
pseudocysts. Abdominal pain, with or without radiation to the back, is the
usual presenting complaint. A palpable, tender mass may be found in the middle
or left upper abdomen. The serum amylase level is elevated in 75% of patients
at some point during their illness and may fluctuate markedly.
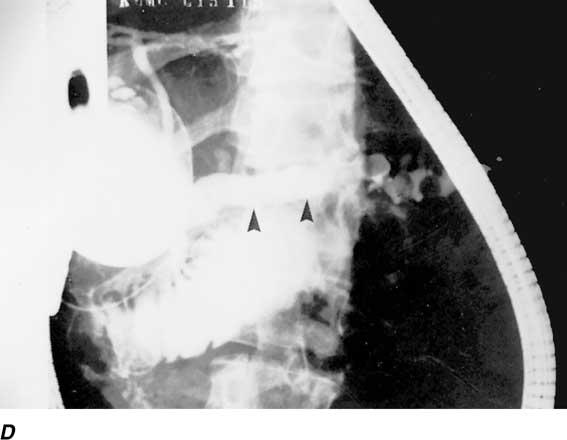
On
x-ray examination, 75% of pseudocysts can be seen to displace some portion of
the gastrointestinal tract (Fig. 304-2). Sonography, however, is reliable in
detecting pseudocysts. Sonography also permits differentiation between an
edematous, inflamed pancreas, which can give rise to a palpable mass, and an
actual pseudocyst. Furthermore, serial ultrasound studies will indicate whether
a pseudocyst has resolved. CT complements ultrasonography in the diagnosis of
pancreatic pseudocyst (Fig. 304-2), especially when the pseudocyst is infected.
|
|
|
|
|
|
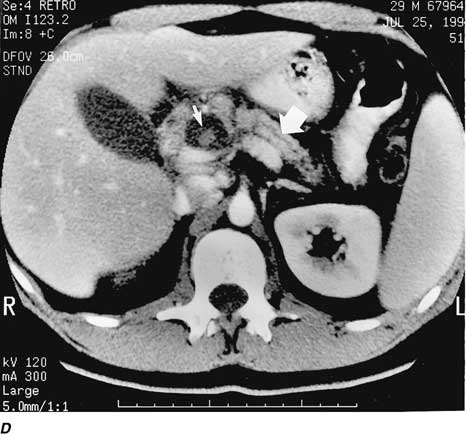
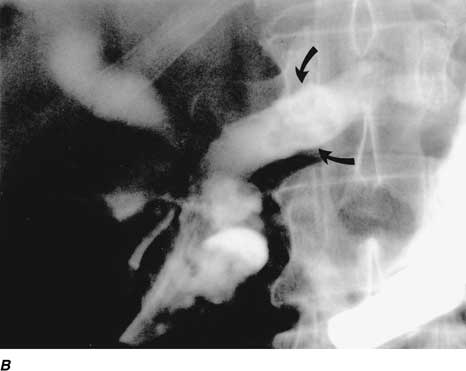
Figure 304-2: Pseudocyst of pancreas. A. Upper gastrointestinal
x-ray showing displacement of stomach by pseudocyst. B. Sonogram showing
pseudocyst (cyst). GB, gallbladder; MVP, portal vein. Behind the large
pseudocyst is seen the calcified head of the pancreas. A dilated common bile
duct (asterisk) is noted. C. CT scan showing pseudocyst. Note the
large, lobulated fluid collection (arrows) surrounding the tail of the
pancreas (arrowheads). Note also the dense, thin rim in the periphery
representing the fibrous capsule of the pseudocyst. D. Spiral CT showing
a pseudocyst (small arrow) with a pseudoaneurysm (light area in
pseudocyst). Note the demonstration of the main pancreatic duct (big arrow),
even though this duct is minimally dilated by ERCP.
In
studies with sonography, pseudocysts were seen to resolve in 25 to 40% of
patients. Pseudocysts that are greater than 5 cm in diameter and that persist
for longer than 6 weeks should be considered for drainage. Recent natural
history studies have suggested that noninterventional, expectant management is
the best course in selected patients with minimal symptoms and no evidence of
active alcohol use in whom the pseudocyst appears mature by radiography and
does not resemble a cystic neoplasm. A significant number of these pseudocysts
resolve spontaneously more than 6 weeks after their formation. Also, these
studies demonstrate that large pseudocyst size is not an absolute indication
for interventional therapy and that many peripancreatic fluid collections
detected on CT in cases of acute pancreatitis resolve spontaneously. A
pseudocyst that does not resolve spontaneously may lead to serious
complications, such as (1) pain caused by expansion of the lesion and pressure
on other viscera, (2) rupture, (3) hemorrhage, and (4) abscess. Rupture of a
pancreatic pseudocyst is a particularly serious complication. Shock almost
always supervenes, and mortality rates range from 14% if the rupture is not
associated with hemorrhage to over 60% if hemorrhage has occurred. Rupture and
hemorrhage are the prime causes of death from pancreatic pseudocyst. A triad of
findings-an increase in the size of the mass, a localized bruit over the mass,
and a sudden decrease in hemoglobin level and hematocrit without obvious
external blood loss-should alert one to the possibility of hemorrhage from a
pseudocyst. Thus, in patients who are stable and free of complications and in
whom serial ultrasound studies show that the pseudocyst is shrinking,
conservative therapy is indicated. Conversely, if the pseudocyst is expanding
and is complicated by rupture, hemorrhage, or abscess, the patient should be
operated on. With ultrasound or CT guidance, sterile chronic pseudocysts can be
treated safely with single or repeated needle aspiration or more prolonged
catheter drainage with a success rate of 45 to 75%. The success rate of these
techniques for infected pseudocysts is considerably less (40 to 50%). Patients
who do not respond to drainage require surgical therapy for internal or
external drainage of the cyst.
Pseudoaneurysms
develop in up to 10% of patients with acute pancreatitis at sites reflecting
the distribution of pseudocysts and fluid collections (Fig. 304-2D). The
splenic artery is most frequently involved, followed by the inferior and
superior pancreatic duodenal arteries. This diagnosis should be suspected in
patients with pancreatitis who develop upper gastrointestinal bleeding without
an obvious cause or in whom thin-cut CT scanning reveals a contrast-enhanced
lesion within or adjacent to a suspected pseudocyst. Arteriography is necessary
to confirm the diagnosis.
Pancreatic Ascites and Pancreatic Pleural
Effusions
Pancreatic
ascites is usually due to disruption of the main pancreatic duct, often by an
internal fistula between the duct and the peritoneal cavity or a leaking
pseudocyst (Chap. 43). This diagnosis is suggested in a patient with an
elevated serum amylase level in whom the ascites fluid has both increased
levels of albumin [>30 g/L (>3.0 g/dL)] and a markedly elevated level of
amylase. The fluid in true pancreatic ascites usually has an amylase
concentration of >20,000 U/L as a result of the ruptured duct or leaking
pseudocyst. Lower amylase elevations may be found in the peritoneal fluid of
patients with acute pancreatitis. In addition, ERCP often demonstrates passage
of contrast material from a major pancreatic duct or a pseudocyst into the
peritoneal cavity. As many as 15% of patients with pseudocysts have concurrent
pancreatic ascites. The differential diagnosis should include intraperitoneal
carcinomatosis, tuberculous peritonitis, constrictive pericarditis, and
Budd-Chiari syndrome.
If
the pancreatic duct disruption is posterior, an internal fistula may develop
between the pancreatic duct and the pleural space, producing a pleural
effusion, which is usually left-sided and often massive. This complication
often requires thoracentesis or chest tube drainage.
Treatment
usually requires the use of nasogastric suction and parenteral alimentation to
decrease pancreatic secretion. In addition, paracentesis is performed to keep
the peritoneal cavity free of fluid and, it is hoped, to effect sealing of the
leak. The long-acting somatostatin analogue octreotide, which inhibits
pancreatic secretion, is useful in cases of pancreatic ascites and pleural
effusion. If ascites continues to recur after 2 to 3 weeks of medical
management, the patient should be operated on after pancreatography to define
the anatomy of the abnormal duct. A disrupted main pancreatic duct can also be
treated effectively by stenting. Patients in whom ERCP identifies two or more
sites of extravasation are unlikely to respond to conservative management
and/or stenting.
Chronic Pancreatitis and Pancreatic Exocrine
Insufficiency
General and Etiologic Considerations
Chronic
inflammatory disease of the pancreas may present as episodes of acute
inflammation in a previously injured pancreas or as chronic damage with
persistent pain or malabsorption. The causes of relapsing chronic pancreatitis
are similar to those of acute pancreatitis (Table 304-1), except that there is
an appreciable incidence of cases of undetermined origin. In addition, the
pancreatitis associated with gallstones is predominantly acute or
relapsing-acute in nature. A cholecystectomy is almost always performed in
patients after the first or second attack of gallstone-associated pancreatitis.
Patients with chronic pancreatitis may present with persistent abdominal pain,
with or without steatorrhea; some (~15%) present with steatorrhea and no pain.
Patients
with chronic pancreatitis in whom there is extensive destruction of the
pancreas (less than 10% of exocrine function remaining) have steatorrhea and
azotorrhea. Among American adults, alcoholism is the most common cause of
clinically apparent pancreatic exocrine insufficiency, while cystic fibrosis is
the most frequent cause in children. In up to 25% of American adults with
chronic pancreatitis, the cause is not known; that is, they have idiopathic
chronic pancreatitis. Mutations of the cystic fibrosis transmembrane
conductance regulator (CFTR) gene have been documented in patients with
idiopathic chronic pancreatitis. It has been estimated that in patients with
idiopathic pancreatitis the frequency of a single CFTR mutation is 11 times the
expected frequency and the frequency of two mutant alleles is 80 times the
expected frequency. The results of sweat chloride testing are not diagnostic of
cystic fibrosis in these patients. However, these patients have functional
evidence of a defect in CFTR-mediated ion transport in nasal epithelium. It is
suggested that up to 25% of patients with idiopathic chronic pancreatitis may
have abnormalities of the CFTR gene. The therapeutic and prognostic implication
of these findings remain to be determined. In other parts of the world, severe
protein-calorie malnutrition is a common cause. Table 304-5 lists other causes
of pancreatic exocrine insufficiency, but they are relatively uncommon.
Table 304-5: Causes of Pancreatic Exocrine
Insufficiency
|
Pathophysiology
The
events that initiate an inflammatory process in the pancreas are still not well
understood, and the many hypotheses will not be reviewed here. In the case of
alcohol-induced pancreatitis, it has been suggested that the primary defect may
be the precipitation of protein (inspissated enzymes) in the ducts. The
resulting ductal obstruction could lead to duct dilation, diffuse atrophy of
the acinar cells, fibrosis, and eventual calcification of some of the protein
plugs. However, the fact that some alcoholic patients with recurrent acute
pancreatitis show no evidence of chronic pancreatitis does not support this
hypothesis. In fact, experimental and clinical observations have shown that
alcohol has direct toxic effects on the pancreas. While patients with alcohol-induced
pancreatitis generally consume large amounts of alcohol, some consume very
little (50 g/d or less). Thus prolonged consumption of "socially
acceptable" amounts of alcohol is compatible with the development of
pancreatitis. In addition, the finding of extensive pancreatic fibrosis in
patients who died during their first attack of clinical acute alcohol-induced
pancreatitis supports the concept that such patients already have chronic
pancreatitis.
Clinical Features
Patients
with relapsing chronic pancreatitis may present with symptoms identical to
those of acute pancreatitis, but pain may be continuous, intermittent, or
absent. The pathogenesis of this pain is poorly understood. Although the
classic description is of epigastric pain radiating through the back, the pain
pattern is often atypical; the pain may be worst in the right or left upper
quadrant of the back or may be diffuse throughout the upper abdomen; it may
even be referred to the anterior chest or flank. Characteristically it is
persistent, deep-seated, and unresponsive to antacids. It often is worsened by
ingestion of alcohol or a heavy meal (especially one rich in fat). Often the
pain is severe enough to necessitate the frequent use of narcotics.
Weight
loss, abnormal stools, and other signs or symptoms suggestive of malabsorption
(see Table 286-5) are common in chronic pancreatitis. However, clinically
apparent deficiencies of fat-soluble vitamins are surprisingly rare. The
physical findings in these patients are usually not impressive, so that there
is a disparity between the severity of the abdominal pain and the physical
signs (other than some abdominal tenderness and mild temperature elevation).
Diagnostic Evaluation
In
contrast to relapsing acute pancreatitis, the serum amylase and lipase levels
are usually not elevated in chronic pancreatitis. Elevations of serum bilirubin
and alkaline phosphatase levels may indicate cholestasis secondary to chronic
inflammation around the common bile duct (Fig. 304-3). Many patients
demonstrate impaired glucose tolerance, and some have an elevated fasting blood
glucose level.
|
|
|
|
|
|
Figure 304-3: Radiologic abnormalities in chronic
pancreatitis. A. Pancreatic calcification (arrows) and stenosis
(tapering) of the intrahepatic portion of the common bile duct demonstrated by
percutaneous transhepatic cholangiography. B. Pancreatic calcification (large
white arrow) demonstrated by sonography. Note dilated pancreatic duct (thin
white arrow) and splenic vein (open arrow). C. Pancreatic
calcification (vertical arrows) and dilated pancreatic duct (horizontal
arrow) demonstrated by CT scan. D. Endoscopic retrograde
cholangiogram shows grossly dilated pancreatic ducts (arrows) in a
patient with long-standing pancreatitis.
The
classic triad of pancreatic calcification, steatorrhea, and diabetes mellitus
usually establishes the diagnosis of chronic pancreatitis and exocrine
pancreatic insufficiency but is found in less than one-third of chronic
pancreatitis patients. Accordingly, it is often necessary to perform an
intubation test such as the secretin stimulation test, which usually
gives abnormal results when 60% or more of pancreatic exocrine function has
been lost. Approximately 40% of patients with chronic pancreatitis have cobalamin
(vitamin B12) malabsorption, which can be corrected by the
administration of oral pancreatic enzymes. There is usually a marked excretion
of fecal fat (Chap. 286), which can be reduced by the administration of oral
pancreatic enzymes. The serum trypsinogen (Chap. 303) and the D-xylose urinary
excretion test are useful in patients with "pancreatic steatorrhea,"
since the trypsinogen level will be abnormal, and D-xylose excretion usually is
normal. A decreased serum trypsinogen level strongly suggests severe pancreatic
exocrine insufficiency.
The
radiographic hallmark of chronic pancreatitis is the presence of scattered
calcification throughout the pancreas (Fig. 304-3). Diffuse pancreatic
calcification indicates that significant damage has occurred and obviates the
need for a secretin test. While alcohol is by far the most common cause,
pancreatic calcification also may be seen in cases of severe protein-calorie
malnutrition, hereditary pancreatitis, posttraumatic pancreatitis,
hyperparathyroidism, islet cell tumors, and idiopathic chronic pancreatitis. A
large prospective study has shown convincingly that pancreatic calcification decreases
or even disappears spontaneously in one-third of patients with severe chronic
pancreatitis; this outcome may also follow ductal decompression. Pancreatic
calcification is a dynamic process that is incompletely understood.
Sonography,
CT, and ERCP greatly aid the diagnosis of pancreatic disease. In addition to
excluding pseudocysts and pancreatic cancer, sonography and CT may show
calcification or dilated ducts associated with chronic pancreatitis (Fig.
304-4). ERCP is the only major technique that provides a direct view of the
pancreatic duct. In patients with alcohol-induced pancreatitis, ERCP may reveal
a pseudocyst missed by sonography or CT.
|
|
|
Figure 304-4: Chronic pancreatitis and pancreatic
calculi: CT scan and ERCP appearance. A. In this contrast-enhanced CT
scan of the abdomen, there is evidence of an atrophic pancreas with multiple
calcifications (arrows). Note the markedly dilated pancreatic duct seen
in this section through the body and tail (open arrows). B. ERCP
in the same patient demonstrates the dilated pancreatic duct as well as an
intrapancreatic duct calculus (arrows). These findings correlate nicely
with the CT scan appearance.
Complications of Chronic Pancreatitis
The
complications of chronic pancreatitis are protean. Cobalamin (vitamin
B12) malabsorption occurs in 40% of patients with
alcohol-induced chronic pancreatitis and in virtually all with cystic fibrosis.
It is consistently corrected by the administration of pancreatic enzymes
(containing proteases). It may be due to excessive binding of cobalamin by
cobalamin-binding proteins other than intrinsic factor, which ordinarily are
destroyed by pancreatic proteases and therefore do not compete with intrinsic
factor for cobalamin binding. Although most patients show impaired glucose
tolerance, diabetic ketoacidosis and coma are uncommon. Similarly,
end-organ damage (retinopathy, neuropathy, nephropathy) is also uncommon, and
the appearance of these complications should raise the question of concomitant
genetic diabetes mellitus. A nondiabetic retinopathy, peripheral in location
and secondary to vitamin A and/or zinc deficiency, is common in these patients.
Effusions containing high concentrations of amylase may occur into the
pleural, pericardial, or peritoneal space. Gastrointestinal bleeding may
occur from peptic ulceration, gastritis, a pseudocyst eroding into the
duodenum, or ruptured varices secondary to splenic vein thrombosis due to
inflammation of the tail of the pancreas. Icterus may occur, caused
either by edema of the head of the pancreas, which compresses the common bile
duct, or by chronic cholestasis secondary to a chronic inflammatory reaction
around the intrapancreatic portion of the common bile duct (Fig. 304-3). The
chronic obstruction may lead to cholangitis and ultimately to biliary cirrhosis.
Subcutaneous fat necrosis may appear as tender red nodules on the lower
extremities. Bone pain may be secondary to intramedullary fat necrosis.
Inflammation of the large and small joints of the upper and lower extremities
may occur. The incidence of pancreatic carcinoma is increased in patients with
chronic pancreatitis who have been followed for 2 or more years. Twenty years
after the diagnosis of chronic pancreatitis, the cumulative risk of pancreatic
carcinoma is 4%. Perhaps the most common and troublesome complication is
addiction to narcotics.
![]() Treatment
Treatment
Therapy
for patients with chronic pancreatitis is directed toward two major problems-pain
and malabsorption. Patients with intermittent attacks of pain are treated
essentially like those with acute pancreatitis (see above). Patients with
severe and persistent pain should avoid alcohol completely and avoid large
meals rich in fat. Since the pain is often severe enough to require frequent
use of narcotics (and hence addiction), a number of surgical procedures have
been developed for pain relief. ERCP allows the surgeon to plan the operative
approach. If there is a stricture of the pancreatic duct, a local resection
may ameliorate the pain. Unfortunately, isolated localized strictures are not
common. In most patients with alcohol-induced disease, the pancreas is
diffusely involved, and surgically correctible localized ductal disease is
rare. When there is primary ductal obstruction and dilation, ductal
decompression may provide effective pain palliation. Short-term pain relief may
be achieved in up to 80% of patients, while long-term pain relief occurs in
approximately 50%. In some of these patients, however, pain relief can be
achieved only by resecting 50 to 95% of the gland. Although pain relief is
achieved in three-quarters of these patients, they tend to develop pancreatic
endocrine and exocrine insufficiency and must be treated with pancreatic enzyme
replacement therapy. It is important to screen patients carefully, for such
radical surgery is contraindicated in those who are severely depressed or
suicidal or who continue to drink. Procedures such as splanchnicectomy, celiac
ganglionectomy, and nerve blocks usually bring only temporary relief and are
not recommended. Endoscopic treatment of chronic pancreatitis may involve
sphincterotomy of the minor or major pancreatic sphincter, dilatation of
strictures, removal of calculi, or stenting of the ventral or dorsal pancreatic
duct. Although many of these techniques are technically impressive, none has
been subjected to a randomized trial in patients with chronic pancreatitis. In
addition, significant complications-acute pancreatitis, pancreatic abscess,
damage to the pancreatic duct, and death-have occurred in up to 36% of patients
after stent placement.
Three
double-blind trials have demonstrated that administration of pancreatic enzymes
decreases abdominal pain in selected patients with chronic pancreatitis. In
these trials, approximately 75% of the patients evaluated experienced pain
relief. The patients most likely to respond are those with mild to moderate
exocrine pancreatic dysfunction, as evidenced by an abnormal secretin test, normal
fat absorption, and minimal abnormalities on ERCP examination. These clinical
observations seem to fit with data from human beings and experimental animals
demonstrating a negative feedback regulation for pancreatic exocrine secretion
controlled by the amount of proteases within the lumen of the proximal small
intestine. It seems reasonable to use the following approach for patients with
severe, persistent, or continuous abdominal pain thought to be caused by
chronic pancreatitis. After other causes of abdominal pain (peptic ulcer,
gallstones, etc.) have been excluded, a pancreatic sonogram should be
done. If no mass is found, a secretin test may be performed, because its
results usually are abnormal in cases of chronic pancreatitis with pain. If the
results are abnormal (i.e., decreased bicarbonate concentration or volume
output), a 3- to 4-week trial of pancreatic enzyme administration is
appropriate. Eight conventional tablets or capsules are taken at meals and at
bedtime. There are a number of studies suggesting that patients may have
small-duct chronic pancreatitis and chronic abdominal pain with a normal
appearance on radiographic evaluations (ultrasound, CT, ERCP) but abnormal
results on hormone stimulation tests (secretin test) and/or abnormal pancreatic
histology. Such minimal-change chronic pancreatitis may respond well to
pancreatic enzyme therapy (non-enteric-coated) for relief of abdominal pain. If
no relief is obtained, and especially if the volume secreted during the
secretin test is very low, ERCP should be performed. If a pseudocyst or a
localized ductal obstruction is found, surgery should be considered. A patient
who has dilated ducts may be a candidate for a surgical ductal decompression
procedure. This procedure provides short-term relief in up to 80% of patients,
although long-term results are closer to 50%. Some studies have shown
octreotide to be effective in decreasing abdominal pain in patients with severe
large-duct disease. If no surgically remediable lesion is found and severe pain
continues despite abstinence from alcohol, subtotal pancreatic resection may be
necessary.
The
treatment of malabsorption rests on the use of pancreatic enzyme replacement
therapy. Diarrhea and steatorrhea are usually improved by this treatment,
although the steatorrhea may not be completely corrected. The major problem is
delivering enough active enzyme into the duodenum. Steatorrhea could be
abolished if 10% of the normal amount of lipase could be delivered to the
duodenum at the proper time. This concentration of lipase cannot be achieved
with the current preparations of pancreatic enzymes, even if the latter are
given in large doses. The reason for these poor results may be that lipase is
inactivated by gastric acid, that food empties from the stomach faster than do
the pancreatic enzymes, and that batches of commercially available pancreatic
extracts vary in enzyme activity.
For
the usual patient, two or three enteric-coated capsules or eight conventional
(non-enteric-coated) tablets of a potent enzyme preparation should be
administered with meals. Some patients using conventional tablets require
adjuvant therapy to improve enzyme replacement treatment. H2
receptor antagonists, sodium bicarbonate, and proton pump inhibitors are
effective adjuvants. Antacids containing calcium carbonate or magnesium
hydroxide are not effective and may actually result in increased steatorrhea.
Several publications have reported colonic strictures in patients with cystic
fibrosis receiving extraordinarily high doses of high-potency pancreatic enzyme
preparations. Such lesions have not been reported in adults with chronic
pancreatitis.
Supportive
measures include diet restriction and pain medications. The diet should be moderate
in fat (30%), high in protein (24%), and low in carbohydrate (40%). Restriction
of long-chain triglyceride intake can help patients who do not respond
satisfactorily to pancreatic enzyme therapy. Use of foods containing mainly
medium-chain fatty acids, which do not require lipase for digestion, may be
beneficial. Nonnarcotic analgesics should be emphasized. Patients taking
narcotic drugs for pain relief often become addicted and continue to have pain.
Patients
with severe exocrine pancreatic insufficiency secondary to alcohol who continue
to drink have a high mortality rate (in one series, 50% of patients who were
followed for 5 to 12 years died during this period) and significant morbidity
(weight loss, lassitude, vitamin deficiency, and narcotic addiction). Chronic
pancreatitis carries significant medical and social costs. A recent study found
that pancreatitis led to retirement in 11% of patients with the disease,
accounting for 45% of all retirements. In 87% of patients with chronic
pancreatitis unable to maintain gainful employment, alcoholism was a
contributing factor. Patients with chronic pancreatitis also use substantial
medical resources. In 1987 in the United States, this diagnosis accounted for
122,000 recorded outpatient visits and 56,000 hospital admissions. Pain may
abate if progressive severe exocrine insufficiency continues. Patients who
abstain from alcohol and use vigorous replacement therapy for
maldigestion-malabsorption do reasonably well.
Hereditary Pancreatitis
Hereditary
pancreatitis is a rare disease that is similar to chronic pancreatitis except
for an early age of onset and evidence of hereditary factors (involving an
autosomal dominant gene with incomplete penetrance). A genome-wide search using
genetic linkage analysis identified the hereditary pancreatitis gene on
chromosome 7. An R117H mutation in the cationic trypsinogen gene occurs in most
of the families with hereditary pancreatitis that have been studied. Molecular
modeling predicts the formation of hydrolysis-resistant trypsin that could lead
to pancreatic autodigestion. These patients have recurring attacks of severe
abdominal pain which may last from a few days to a few weeks. The serum amylase
and lipase levels may be elevated during acute attacks but usually are normal.
Patients frequently develop pancreatic calcification, diabetes mellitus, and
steatorrhea, and, in addition, they have an increased incidence of pancreatic
carcinoma. Such patients often require ductal decompression for pain relief.
Abdominal complaints in relatives of patients with hereditary pancreatitis
should raise the question of pancreatic disease.
Other Conditions
Annular Pancreas
When
the ventral pancreatic anlage fails to migrate correctly to make contact with
the dorsal anlage, the result may be a ring of pancreatic tissue encircling the
duodenum. Such an annular pancreas may cause intestinal obstruction in the
neonate or the adult. Symptoms of postprandial fullness, epigastric pain,
nausea, and vomiting may be present for years before the diagnosis is
entertained. The radiographic findings are symmetric dilation of the proximal
duodenum with bulging of the recesses on either side of the annular band,
effacement but not destruction of the duodenal mucosa, accentuation of the
findings in the right anterior oblique position, and lack of change on repeated
examinations. The differential diagnosis should include duodenal webs, tumors
of the pancreas or duodenum, postbulbar peptic ulcer, regional enteritis, and
adhesions. Patients with annular pancreas have an increased incidence of
pancreatitis and peptic ulcer. Because of these and other potential
complications, the treatment is surgical even if the condition has been present
for years. Retrocolic duodenojejunostomy is the procedure of choice, although
some surgeons advocate Billroth II gastrectomy, gastroenterostomy, and
vagotomy.
Pancreas Divisum
Pancreas
divisum occurs when the embryologic ventral and dorsal pancreatic anlagen fail
to fuse, so that pancreatic drainage is accomplished mainly through the
accessory papilla. Pancreas divisum is the most common congenital anatomic
variant of the human pancreas. Current evidence indicates that this anomaly
does not predispose to the development of pancreatitis in the great majority of
patients who harbor it. However, the combination of pancreas divisum and a
small accessory orifice could result in dorsal duct obstruction. The challenge
is to identify this subset of patients with dorsal duct pathology. Cannulation
of the dorsal duct by ERCP is not as easily done as is cannulation of the
ventral duct. Patients with pancreatitis and pancreas divisum demonstrated by
ERCP should be treated with conservative measures. In many of these patients,
pancreatitis is idiopathic and unrelated to the pancreas divisum. Endoscopic or
surgical intervention is indicated only when the above methods fail. If marked
dilation of the dorsal duct can be demonstrated, surgical ductal decompression
should be performed. The appropriate therapy for patients without dilation of
the dorsal duct is not yet defined. It should be stressed that the ERCP
appearance of pancreas divisum-i.e., a small-caliber ventral duct with an
arborizing pattern-may be mistaken as representing an obstructed main
pancreatic duct secondary to a mass lesion.
Macroamylasemia
In
macroamylasemia, amylase circulates in the blood in a polymer form too large to
be easily excreted by the kidney. Patients with this condition demonstrate an
elevated serum amylase value, a low urinary amylase value, and a Cam/Ccr
ratio of less than 1%. The presence of macroamylase can be documented by
chromatography of the serum. The prevalence of macroamylasemia is 1.5% of the
nonalcoholic general adult hospital population. Usually macroamylasemia is an
incidental finding and is not related to disease of the pancreas or other
organs.
Macrolipasemia
has now been documented in a few patients with cirrhosis or non-Hodgkin's
lymphoma. In these patients, the pancreas appeared normal on ultrasound and CT
examination. Lipase was shown to be complexed with immunoglobulin A. Thus, the
possibility of both macroamylasemia and macrolipasemia should be
considered in patients with elevated blood levels of these enzymes.